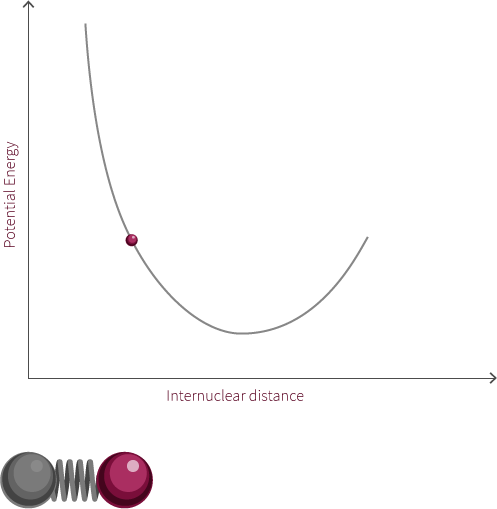
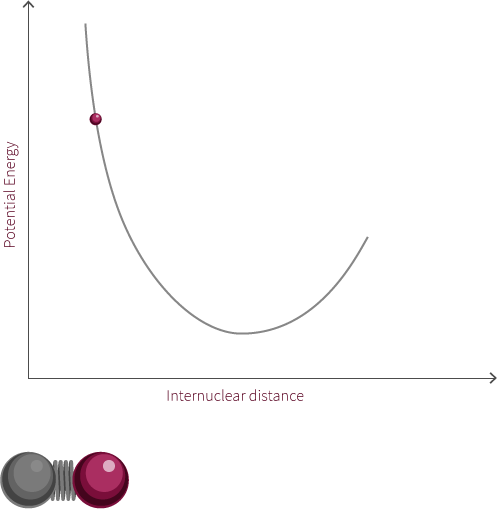
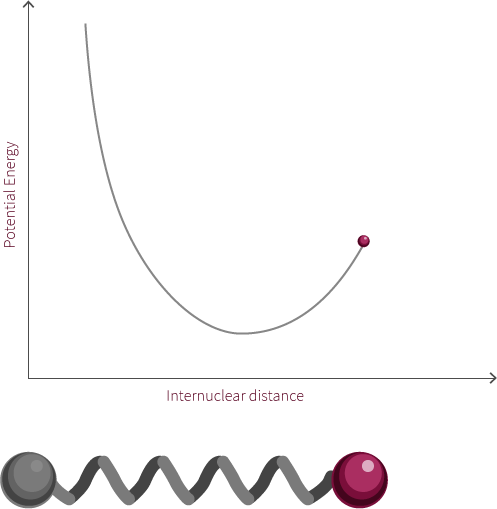
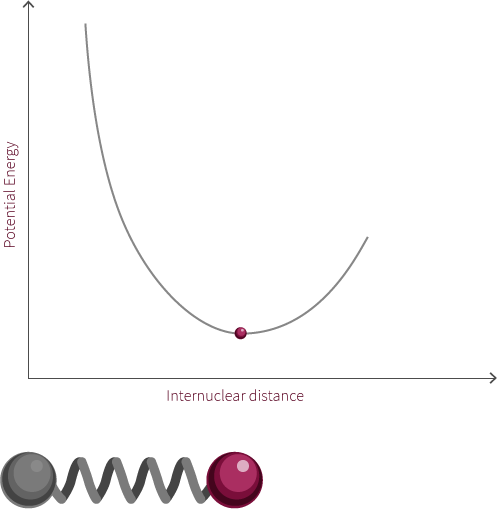
The system to the right is not a true simple harmonic oscillator. However, it does show oscillatory motion under certain conditions. The system to the right follows the curve of a Morse potential.
morse potential
Morse potentials are used to model the interaction between two atoms in a diatomic molecule. Atoms oscillate back and forth due to the interplay of forces acting upon the atoms. There is a strong repulsive force that exists when the two atoms are close to one another. This is due to electrostatic repulsion between the two positively charged nuclei as well as the repulsion between inner-orbital electorns. The attractive force becomes the dominant force as the distance increases. If the distance is too great, there is no longer an electric field interaction between the atoms. The energy at this distance corresponds to the dissociation energy of the bond.
In this example of an oscillating system, we have two spheres which exert both a repulsive and attractive force on each other. In this proposed system, repulsive forces are stronger than attractive forces when the spheres are close to each other. The net force, shows the summation of the two forces and how this changes with the distance between the spheres.
balls and arrows
There exists within this system an equilibrium distance where the net force is equal to zero. When the system is disturbed, it will oscillate around this equilibrium position.
Try pushing the spheres together or pulling the spheres apart. Observe the relationship between the two spheres and changes to potential energy. What else do you notice? Try moving the spheres as far away from each other as possible. What happens?